Shiroh Futaki
Kyoto University
Talk Title
Liquid Droplet Formation and Facile Cytosolic Translocation of IgG in the Presence of Attenuated Cationic Amphiphilic Lytic Peptides
Presentation Time
SESSION 5: PEPTIDE MATERIALS AND DELIVERY
Monday, June 26, 2023, at 11:55 am - 12:15 pm
We previously reported an attenuated cationic amphiphilic lytic, ACAL, peptide, engineered from a natural hemolytic peptide, named L17E.1 Substantial cytosolic delivery of biomacromolecules, including immunoglobulin G (IgG), was attained in the presence of this peptide. Fc region binding peptide conjugated with L17E trimer [FcB(L17E)3] was designed for more efficient IgG delivery into cells,2 see figure.
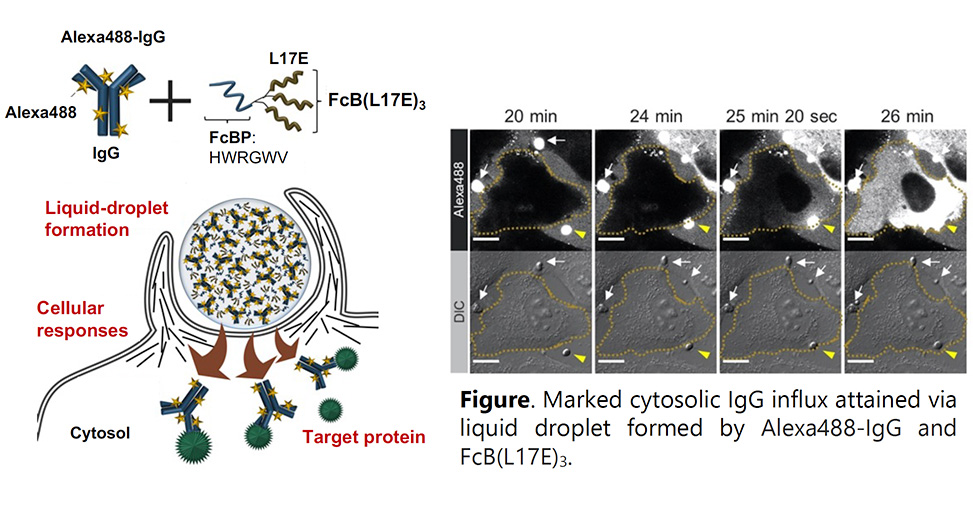
Particle-like liquid droplets were generated by mixing Alexa Fluor 488 labeled IgG (Alexa488-IgG) with FcB(L17E)3. Droplet contact with the cellular membrane led to spontaneous influx and distribution of Alexa488-IgG throughout cells in serum containing medium. Involvement of cellular machinery accompanied by actin polymerization and membrane ruffling was suggested for the translocation. Alexa488-IgG negative charges were crucial in liquid droplet formation with positively charged FcB(L17E)3.
Successful intracellular delivery of Alexa Fluor 594-labeled anti-nuclear pore complex antibody allowed binding to cellular targets in the presence of FcB(L17E)3. Conjugation of L17E to hydrophilic polymers was also found to provide a similar mode of cytosolic IgG delivery, suggesting design flexibility for intracellular delivery via liquid droplet, or coacervate.
References
1. Misao Akishiba, Toshihide Takeuchi, Yoshimasa Kawaguchi, Kentarou Sakamoto, Hao-Hsin Yu, Ikuhiko Nakase, Tomoka Takatani-Nakase, Fatemeh Madani, Astrid Gräslund & Shiroh Futaki. Cytosolic Antibody Delivery by Lipid-Sensitive Endosomolytic Peptide. Nat. Chem., 2017, 9, 751-761.
2. Takahiro Iwata, Hisaaki Hirose, Kentarou Sakamoto, Yusuke Hirai, Jan Vincent V. Arafiles, Misao Akishiba, Miki Imanishi, Shiroh Futaki. Liquid Droplet Formation and Facile Cytosolic Translocation of IgG in the Presence of Attenuated Cationic Amphiphilic Lytic Peptides. Angew. Chem. Int. Ed., 2021, 60, 19804-19812.
Professor Shiroh Futaki graduated from Faculty of Pharmaceutical Sciences, Kyoto University in 1983 and obtained his Ph.D. in Pharmaceutical Sciences in 1989 from Kyoto University under the supervision of Professors Haruaki Yajima and Nobutaka Fujii. Meanwhile he was appointed as a Research Associate at Faculty of Pharmaceutical Sciences at the University of Tokushima in 1987. His postdoctoral training was with Professor James Manning at the Rockefeller University, New York City, 1989-1991. He was promoted to Associate Professor at Faculty of Pharmaceutical Sciences, The University of Tokushima in 1993, and moved to Institute of Chemical Research, Kyoto University as an Associate Professor in 1997, where he was promoted to a Full Professor of Biochemistry in 2005. He was also appointed as a PREST and SORST Investigator of JST from 2003 to 2006, and from 2006 to 2008, respectively. He visited Université Pierre et Marie Curie, Paris as a professeur invité in 2010. Professor Futaki is now a Trustee of The Japanese Peptide Society and also an Associate Editor of Molecular Therapy and a member in the Editorial Boards of four scientific journals. His honors include The Japanese Peptide Society Award for Young Scientists, 1997, Daiichi Pharmaceutical Award in Synthetic Organic Chemistry, Japan, 1997, and The Pharmaceutical Society of Japan Award for Young Scientists, 1998.
Research in the Futaki lab focuses on development of cell-penetrating peptide vectors and elucidation of the internalization mechanisms. Arginine-rich cell-penetrating peptides are an attractive means of intracellular delivery of therapeutic molecules. This has also potential in chemical and biological studies on cellular functions. Using designed peptides as an analytical tool, the group is exploring the methods of internalization of arginine-rich peptides and developing novel efficient and effective means for intracellular delivery.






