Lara Malins
Research School of Chemistry at the Australian National University
Talk Title
Dialing in the Potential: Tunable Electrochemical Peptide Modifications
Presentation Time
EARLY CAREER LECTURESHIP AWARD
Thursday, June 29, 2023, at 11:30 pm - 12:00 pm
The use of electricity to drive chemical reactions is an appealing approach to green and sustainable synthesis. Moreover, electrochemistry provides powerful opportunities for the precise control of chemical reactivity by enabling practitioners to "dial in" the potential or current at which a reaction is performed. For highly functionalized molecules, this tunability provides unprecedented opportunities for targeted chemical modifications.1
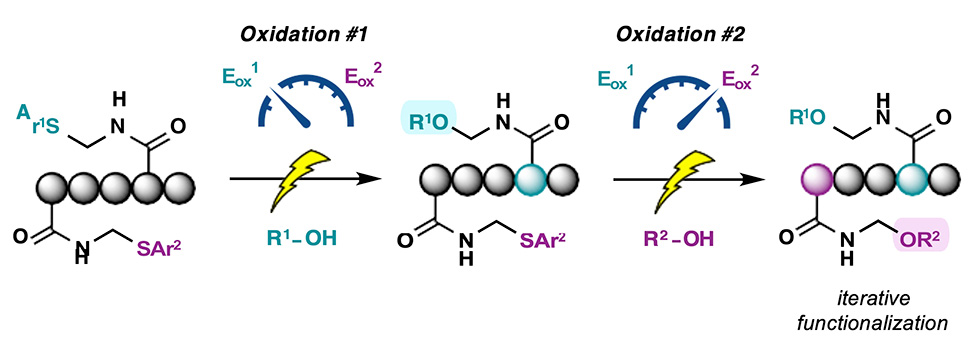
Despite these appealing characteristics, there are remarkably few examples in which electrochemistry is applied to the tunable functionalization of peptides.1 In this presentation, a method for the selective, iterative electrochemical modification of peptides is presented.2 Designer glutamine residues adorned with discrete "electroauxiliaries" — functional groups electronically-predisposed to anodic oxidation — are incorporated into peptides and exploited for iterative modifications to afford a library of high-value peptide N,O-acetals. The utility of the method is demonstrated on unprotected peptides and bioactive substrates and is applied in a novel approach to peptide macrocyclization.
This targeted approach to electrochemical activation serves to unlock a new level of orthogonality in peptide synthesis, and the strategy has promising applications for the preparation of peptide libraries, including for therapeutic development.
References
1. Mackay, A. S., Payne, R. J., Malins, L. R. Electrochemistry for the Chemoselective Modification of Peptides and Proteins. J. Am. Chem. Soc. 2022, 144, 23.
2. Karipal Padinjare Veedu, D., Connal, L. A., Malins, L. R. Tunable Electrochemical Peptide Modifications: Unlocking New Levels of Orthogonality for Side-Chain Functionalization Angew. Chem. Int. Ed, 2023, 62, e202215470.
Dr. Lara Malins is an Associate Professor at the Research School of Chemistry at the Australian National University, ANU. She completed her B.A. in chemistry at Boston University in 2009 before relocating to The University of Sydney to undertake her PhD with Professor Richard Payne on the development of chemoselective peptide ligation strategies. In 2015, Lara joined the laboratory of Professor Phil Baran at The Scripps Research Institute in La Jolla as a National Institutes of Health Ruth L. Kirschstein postdoctoral research fellow, where she worked on methods for the late-stage synthetic modification of peptides. She returned to Australia in November 2017 to begin her independent career at the ANU, where she joined the Research School of Chemistry as a Lecturer and Australian Research Council, ARC, Discovery Early Career Research Fellow. She is currently a Westpac Research Fellow and Chief Investigator in the ARC Centre of Excellence for Innovations in Peptide and Protein Science, CIPPS.
The Malins lab focuses on the development of new synthetic methods for chemical biology, drug discovery, and natural product synthesis. The group’s interest in leveraging underutilized modes of chemical activation, including electricity, light, and molecular strain, has led to the development of new strategies for the targeted functionalization of peptides, including highly tunable electrochemical oxidations and strain-promoted cycloaddition reactions. Dr. Malins has been recognized as an outstanding early career researcher as a recipient of the Australian Peptide Association’s John Wade Early Career Researcher Award and the Royal Australian Chemical Institute’s Rennie Medal.
2023 Early Career Lectureship Presentation







