Alexander L. Nielsen
École Polytechnique Fédérale de Lausanne
Talk Title
Assessing the Cellular Permeability of Peptidic Macrocycles in High-Throughput
Presentation Time
Dr. Elizabeth Schram YI Oral Competition
Wednesday, June 28, 2023, at 04:15 pm - 04:30 pm
Small cyclic peptides provide an attractive modality for drug development due to their ability to bind challenging targets and their potential to cross membranes for reaching intracellular proteins. In our laboratory, we have recently developed methods to synthesize and screen large combinatorial libraries of small cyclic peptides.1, 3 For example, "m" short linear peptides containing thiol groups at both ends were combinatorially cyclized with "n" bis-electrophilic linker reagents to obtain m × n cyclic peptides that were screened in microwell plates as crude products. While the approaches yielded ligands to several disease targets, not all of them were membrane permeable. A full picture of the membrane permeability of the newly developed format of peptidic macrocycles, and factors that determine their permeability, was lacking.
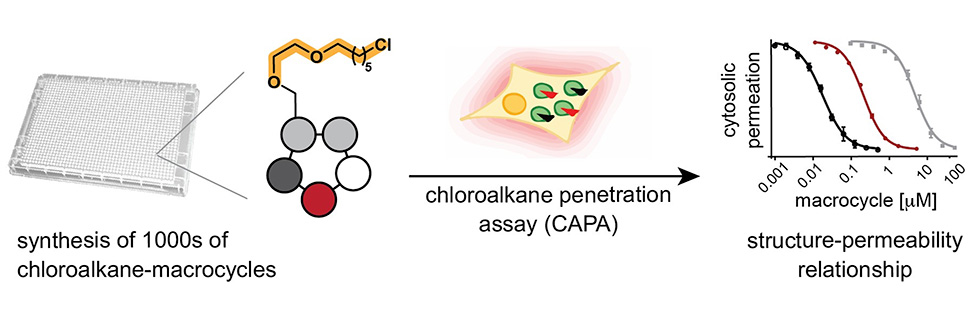
In this work in progress, we have taken advantage of the chloroalkane permeation assay, CAPA, that has recently emerged as a robust method to determine cytosolic permeability of chloroalkane-tagged biomolecules.4 We have established a method to synthesize thousands of diverse chloroalkane-tagged peptidic macrocycles to determine their cytosolic permeability using CAPA. This has given us a new insight into the structure-permeability relationships of an unprecedented number of macrocycles and provides a clearer picture of what features govern permeability of macrocyclic compounds in cellular systems.
References
1. S. S. Kale, M. Bergeron-Brlek, Y. Wu, M. G. Kumar, M. V. Pham, J. Bortoli, J. Vesin, X.-D. Kong, J. Franco Machado, K. Deyle, P. Gonschorek, G. Turcatti, L. Cendron, A. Angelini, And C. Heinis. Thiol-to-Amine Cyclization Reaction Enables Screening of Large Libraries of Macrocyclic Compounds and the Generation of Sub-Kilodalton Ligands. Sci. Adv. 2019, 5, eaaw2851.
2. Sevan Habeshian, Manuel Leonardo Merz, Gontran Sangouard, Ganesh Kumar Mothukuri, Mischa Schüttel, Zsolt Bognár, Cristina Díaz-Perlas, Jonathan Vesin, Julien Bortoli Chapalay, Gerardo Turcatti, Laura Cendron, Alessandro Angelini & Christian Heinis. Synthesis and Direct Assay of Large Macrocycle Diversities by Combinatorial Late-Stage Modification at Picomole Scale. Nat. Commun. 2022, 13, 3823.
3. Zsolt Bognar, Ganesh K. Mothukuri, Alexander L. Nielsen, Manuel L. Merz, Peter M. F. Pânzar, and Christian Heinis. Solid-Phase Peptide Synthesis on Disulfide-Linker Resin Followed by Reductive Release Affords Pure Thiol-Functionalized Peptides. Org. Biomol. Chem. 2022, 20, 5699–5703.
4. Leila Peraro, Kirsten L. Deprey, Matthew K. Moser, Zhongju Zou, Haydn L. Ball, Beth Levine, and Joshua A. Kritzer. Cell Penetration Profiling Using the Chloroalkane Penetration Assay. J. Am. Chem. Soc. 2018, 140, 11360–11369.
Alexander L. Nielsen is a senior postdoctoral researcher in Professor Christian Heinis' lab at EPFL in Lausanne, Switzerland. His research interest revolves around high-throughput experimentation and the synthesis/screening of cyclic peptide libraries against challenging drug targets.
Alexander earned his Ph.D. from University of Copenhagen, 2021, where he worked on design, synthesis and pharmacological characterization of peptidic inhibitors against sirtuin enzymes. During his Ph.D., he was also awarded a Fulbright scholarship, which took him to Scripps Research in La Jolla, CA, to work on covalent inhibitors of Botulinum Neurotoxin with Professor Kim Janda. Outside of the laboratory, he enjoys spending time in the surrounding Swiss mountains.






