Christoph Nitsche
Australian National University
Talk Title
Biocompatible Peptide Cyclisation and Protein Modification
Presentation Time
SESSION 7: FROM NATURAL PRODUCTS TO UNNATURAL BIOACTIVE PEPTIDES
Tuesday, June 27, 2023, at 09:20 am - 09:40 am
Constrained and modified peptides fill an underexplored area of chemical space between small molecule therapies and larger antibodies. Noncanonical modifications, such as cyclisation or stapling, can i enhance metabolic stability by greater resistance towards proteolysis, ii promote biological uptake across cell membranes, and iii decrease the entropic penalty of binding by locking the peptide in the active conformation.
We developed various unnatural amino acids functionalized with cyanopyridine and 1,2-aminothiol groups which can be directly incorporated into peptides.1, 2 Cyclisation and stapling reactions proceed under biocompatible conditions in presence of protein drug targets. Using these approaches in small screening campaigns, we were able to identify various macrocyclic peptide inhibitors of viral proteases. In addition, these amino acids can be incorporated into proteins enabling selective protein modification.
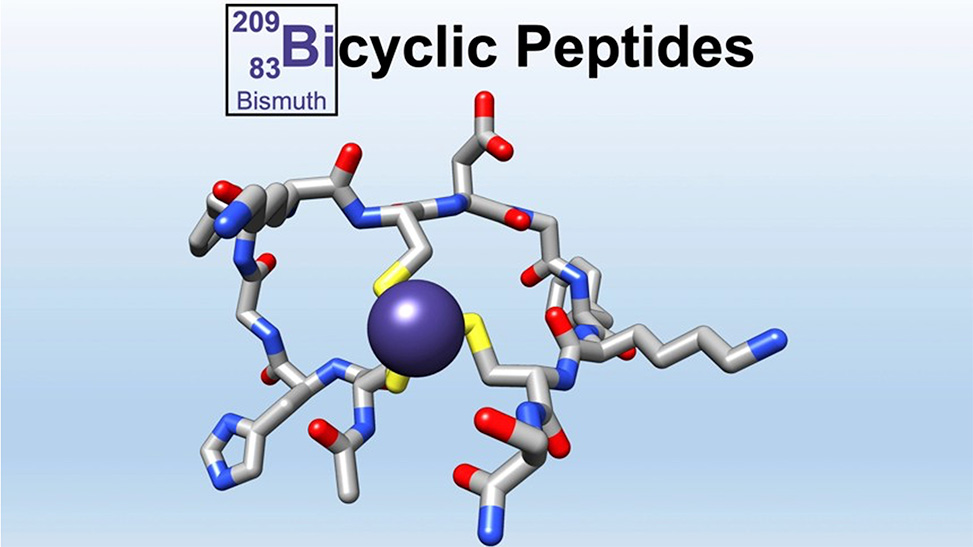
Bicyclic peptides offer even greater conformational rigidity, metabolic stability, and antibody-like affinity and specificity. We explored the reaction between 1,2-aminothiols and 2,6-dicyanopyridine to establish a biocompatible, selective, and catalyst-free pathway to access bicyclic peptides, which displayed plasma stability, conformational preorganization, and high target affinity.3 We also introduced bismuth as a selective, stable, rigid, and green reagent for peptide modification. Bismuth represents the smallest "scaffold" ever explored and allows in situ access to bicyclic peptides for biochemical screening assays.4
References
1. Richard Morewood and Christoph Nitsche. A Biocompatible Stapling Reaction for in situ Generation of Constrained Peptides. Chem. Sci., 2021, 12, 669–674
2. Richard Morewood and Christoph Nitsche. Bioinspired Peptide Stapling Generates Stable Enzyme Inhibitors. Chem. Commun., 2022, 58, 10817–10820.
3. Ullrich, S., George, J., Coram, A. E., Morewood, R., Nitsche, C. Biocompatible and Selective Generation of Bicyclic Peptides. Angew. Chem. Int. Ed., 2022, 61, e202208400
4. Voss, S., Rademann, J., Nitsche, C. Peptide–Bismuth Bicycles: In Situ Access to Stable Constrained Peptides with Superior Bioactivity. Angew. Chem. Int. Ed., 2022, 61, e202113857
Christoph studied chemistry and business administration. He completed his Ph.D. at Heidelberg University in 2014 for which he received the Ph.D. prize for Medicinal Chemistry by the German Chemical Society. He also received a Feodor Lynen Research Fellowship by the Alexander von Humboldt Foundation to work at the Australian National University, ANU, from 2015 to 2018.
After a short period as Rising Star Fellow at the Free University of Berlin, he returned to the ANU with an ARC Discovery Early Career Research Award, 2019. He was appointed Senior Lecturer in 2020 and promoted to Associate Professor in 2023.
Christoph is the recipient of an ARC Future Fellowship, 2022, the John Wade Early Career Researcher Award by the Australian Peptide Association, 2022, the Peter Schwerdtfeger Award by the Australian Association of von Humboldt Fellows, 2022, the Australian Research Award as top researcher in the field of Medicinal Chemistry, 2023, and the Thieme Chemistry Journals Award, 2023.






