Ron Raines
Massachusetts Institute of Technology
Talk Title
A New Role for Methionine Residues
Presentation Time
SESSION 4: PROTEIN ARCHITECTURES - STRUCTURE AND FUNCTION
Monday, June 26, 2023, at 08:30 am - 08:55 am
Cysteine and methionine are relatively rare residues that are well known for their susceptibility to oxidation by reactive oxygen species, ROS. Histidine is the most common residue in enzymic active sites and also reacts with ROS, forming 2-oxo-histidine. The active site of RNase 1, which is a human homologue of RNase A, contains two histidine residues that perform acid–base catalysis to effect RNA cleavage in the oxygen-rich extracellular environment.
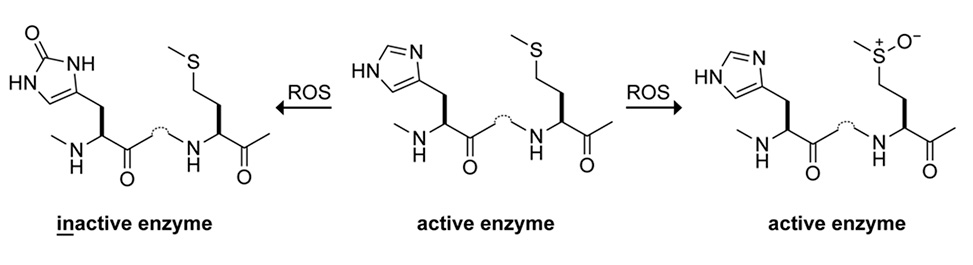
We observed that RNase 1 contains five methionine residues, each proximal to the active-site histidines. We reasoned that these methionine residues could serve as sacrificial "anti-oxidants" that preserve the integrity of the histidines. We found that the susceptibility of RNase 1 to oxidation does indeed increase upon treatment with an oxaziridine –which forms an S=N bond– or replacement of methionine residues with norleucine. Moreover, we find that S–aryl interactions, which can make methionine residues more, or less, susceptible to oxidation, conspire in the anti-oxidant activity.
Acknowledgements
This work was supported by NIH grants R35 GM148220 and R01 CA07308.
The amino-acid sequence of a protein encodes its three-dimensional structure, and this structure manifests itself in biological function. Using techniques that range from synthetic chemistry to cell biology, the Raines group is illuminating in atomic detail both the chemical basis and the biological purpose for protein structure and protein function. The efforts of the Raines group are leading to insights into the relationship between amino-acid sequence and protein function (or dysfunction), as well as to the creation of novel molecules with desirable properties. For example, they have discovered an RNA-cleaving enzyme that is in a multi-site human clinical trial as an anti-cancer agent; revealed that unappreciated forces—the n-to-pi* interaction and C5 hydrogen bond—stabilize all proteins; created hyperstable and human-scale synthetic collagens; and developed processes to synthesize proteins, catalyze their folding, and facilitate their entry into human cells, and to convert crude biomass into useful fuels and chemicals.
The research projects in the Raines laboratory are designed to reveal how biological phenomena can be explained with the principles of chemistry. The hypotheses are far-reaching, and testing them requires the use of techniques and ideas from diverse disciplines. This broad/deep training is appropriate for scientists who want to perform innovative and meaningful research at the widening interface between chemistry and biology.






