Nicholas Sawyer
Fordham University
Talk Title
A Rational Crosslinking Approach for Peptide Polyproline II Helices
Presentation Time
SESSION 1: PEPTIDE TOOLS & PROBES
Sunday, June 25, 2023, at 09:25 am - 09:45 am
Polyproline II helices are ubiquitous structures in proteins with significant enrichment in both structural proteins, for example, collagen, and as ligands for modular protein-protein interaction domains, for example, SH3, WW, and EVH1. Thus, approaches to stabilize peptide polyproline II helices offer opportunities for advanced materials and enhanced modulators of protein-protein interactions. Here we describe a rational approach to stabilize peptide polyproline II helices using site-specific crosslinking.
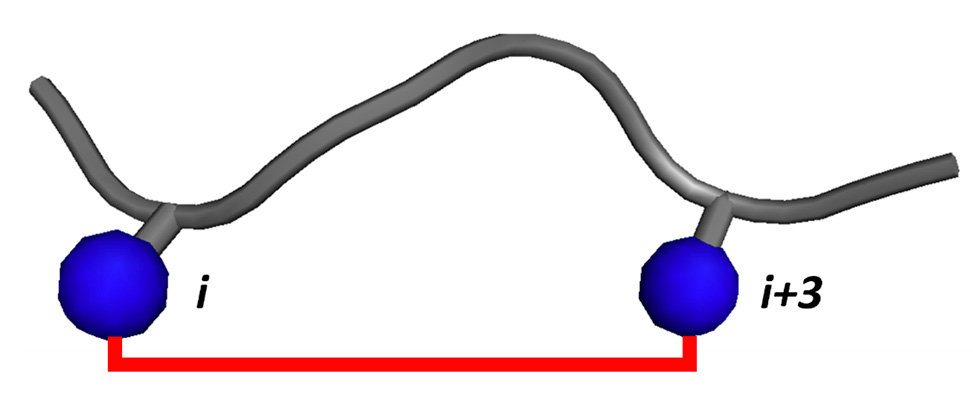
Initial crosslinker design was inspired by a correlation between the number of crosslinker atoms and crosslinked distance across a variety of crosslinkers used to stabilize α-helices. Our model system was the EphB2 receptor-specific SNEW peptide, which adopts a polyproline II helix within its C-terminal half when bound to the receptor. We identified several crosslinked SNEW variants with enhanced inhibitory potency against the EphB2-ephrin B2 interaction, as expected for crosslinker stabilization of the bound peptide structure. These variants also displayed enhanced resistance to protease digestion and retained their specificity for inhibition of the EphB2 receptor over other Eph receptors. Structural analysis of the crosslinked peptide region was consistent with a polyproline II helix conformation.
We envision that this crosslinking approach will be generalizable to stabilize peptides in the polyproline II helical conformation for various biomedical and materials applications.
Research in the Sawyer Group aims to synthesize and evaluate molecules that imitate interacting protein surfaces to investigate the sequence-structure-function relationship of protein-protein interactions involved in human health and disease. Such molecules have utility as both probes for understanding essential biological processes and for innovation in drug development.
Protein-protein interactions, PPIs, are fundamental to biological processes that allow cells to communicate and adapt to stress. The functional groups and their relative orientation on a protein’s surface ultimately determine how strongly and specifically a protein binds to its partner protein/s through the formation of many weak non-covalent interactions. Some functional groups contribute more strongly to complex formation than others and are thus considered "hot spots."
A central hypothesis in the Sawyer Group's research is that molecules that reproduce the hot spots of a protein surface can competitively modulate its PPI functions. This hypothesis has been explored previously for many of the >500,000 PPIs, specifically those that are dominated by regular secondary and tertiary structures, including α-helices, β-sheets, and coiled coils. However, the other half of the protein-protein interactome is mediated by non-regular “loop” structures and remains poorly understood.
At the interface between chemistry and biology, research in the Sawyer Lab lab seeks to develop and apply synthetic approaches to examine the roles of loops in PPIs. Specific research areas include 1) sequence- independent strategies to stabilize specific loop structures and 2) control of loop structure to drive preferential binding to specific protein targets. Ultimately, the synthetic strategies and molecules will provide important insights into fundamental biological processes associated with human health and disease.



